Analytical Ultracentrifugation was invented in the 1920s by the Swede Thé Svedberg. He was the first to equip a rapidly rotating centrifuge with an optical system in order to monitor sedimenting particles. In recognition of this groundbreaking innovation, the sedimentation coefficient was later given the unit Svedberg [S]. He was awarded the Nobel prize for chemistry in 1926 – only 18 years after being promoted to PhD.
Already in 1924, he discovered that “most probably, hemoglobin in solution exhibits a molecular mass of 66800 – in four subunits of 16700.” An early and spectacular success of Analytical Ultracentrifugation.


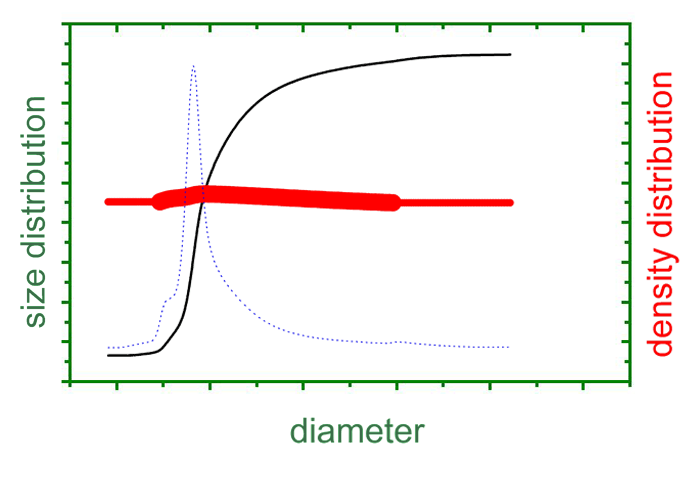
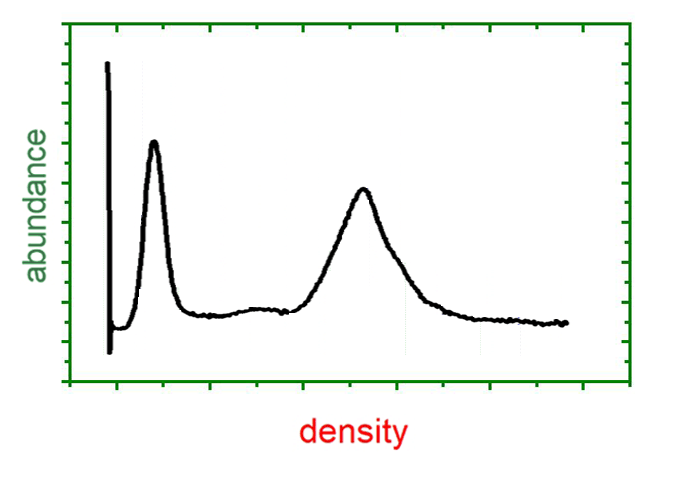
By means of Analytical Ultracentrifugation, Svedberg determined particle size distributions of gold sols. Subsequently, the method was widely applied in biochemistry for the determination of molar masses and sedimentation coefficients. In the 1970s, it was commonly replaced by other methods (light scattering, size exclusion chromatography, gel electrophoresis), but with emerging colloid sciences during the last decade and a newly growing interest in biochemical interactions, new fields of application fields have arisen in addition to traditional applications.
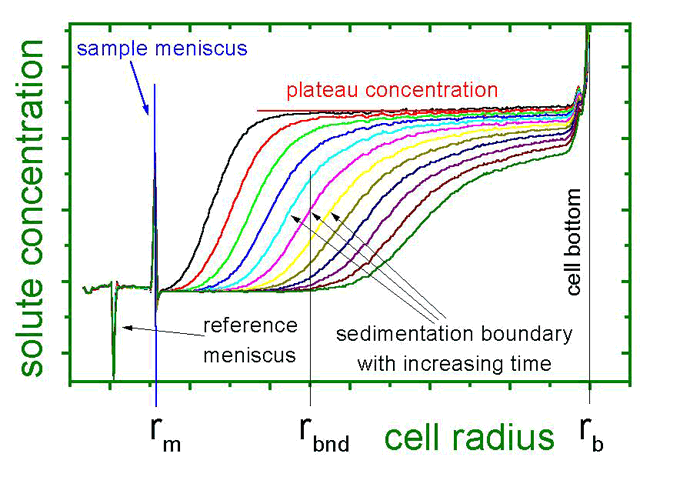
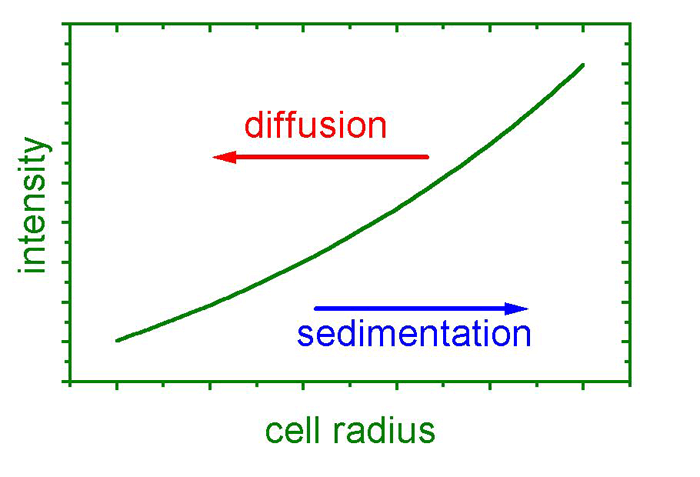
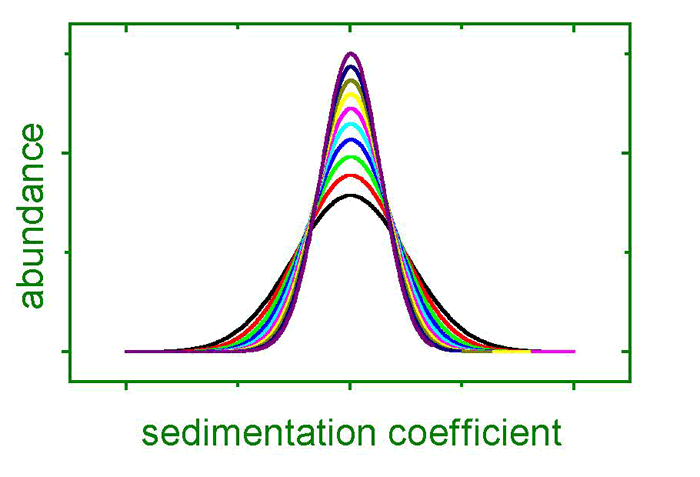
A particle in a sample cell is exposed to centrifugal fields up to the 260,000-fold of Earth’s gravity by spinning the cell at an angular velocity of 60,000 rpm, which is currently the machine’s limit. These fields are sufficient to force particles as small as several nanometers in diameter – or molecules as small as 2,000 Da – to sediment during several hours or less. During the sedimentation process, the concentration profiles in the sample cell are registered by optical detectors, providing time- and locally resolved information. These data yield information on
- mass – a large stone will sink more rapidly in water than a small one
- density – a ball made of steel will sink more rapidly than one of plastic
- shape – a ball will sink more rapidly than a coil, rod or disk.
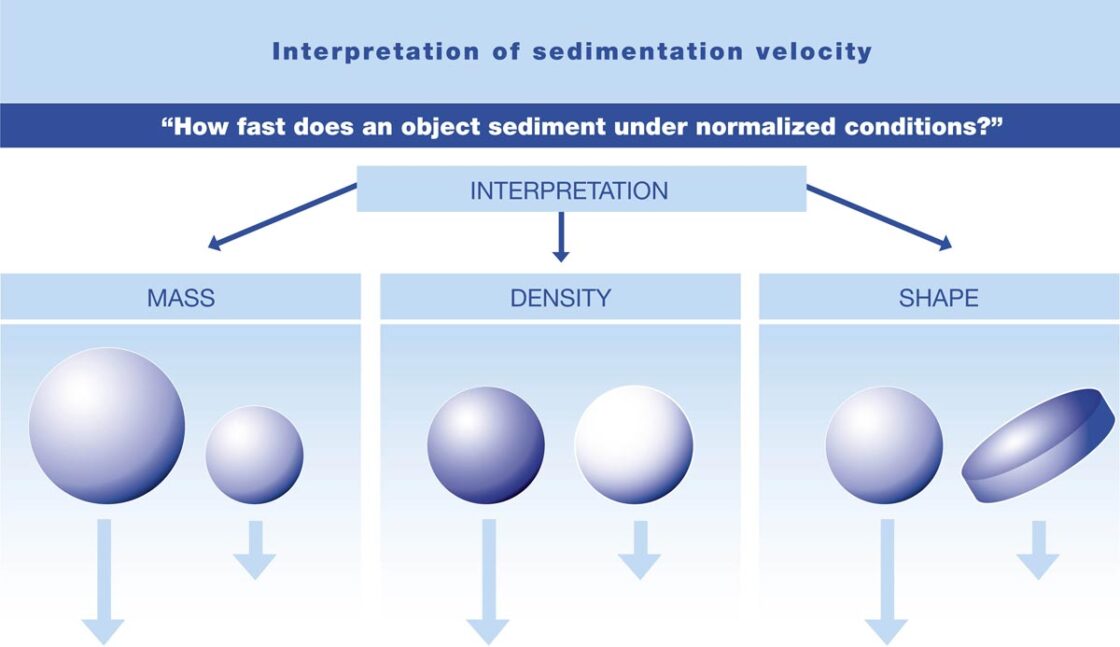
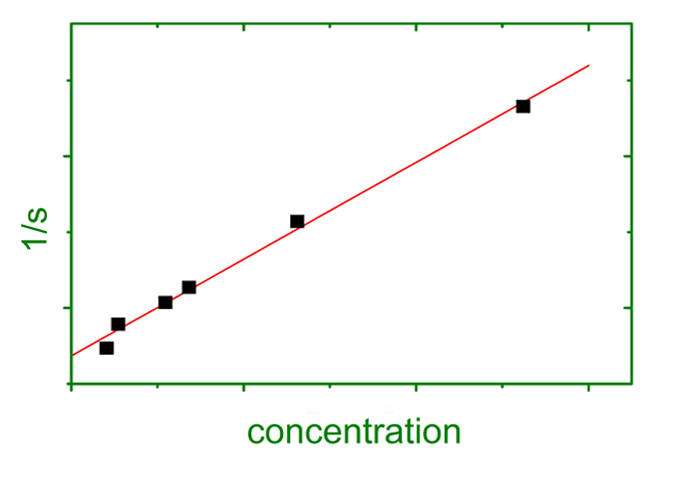
These are the three basic properties that govern an object’s sedimentation velocity. Additionally, concentration and diffusion effects oppose sedimentation, where the latter is time dependent.
Analytical Ultracentrifugation is the only method in colloid analysis that is dependent on mass as the fundamental physical property. In contrast to this, scattering methods are sensitive to changes in the refractive index, rheometry and viscometry are sensitive to shape and interparticle interactions, osmometry solely to the number of particles, other methods to charges. The Analytical Ultracentrifuge occupies a key position due to its most important features: It is dispersive and an absolute method – uniquely based on the physical principle that accelerated mass exhibits a force.
Advantages of Analytical Ultracentrifugation (AUC)
The following features make Analytical Ultracentrifugation such a valuable tool in colloid and biophysical analysis:
- Analytical Ultracentrifugation is an absolute method.
- Analytical Ultracentrifugation is a dispersive method; mixtures are fractionated during analysis.
- Analytical Ultracentrifugation gives access to geometric (size, shape, structure) and thermodynamic properties (equilibrium constants, free energies, enthalpies, entropies).
- With a maximum angular velocity of 60,000 rpm, equivalent to Earth’s gravity 260,000-fold, Analytical Ultracentrifugation is applicable for a particle size range from 1 to 1000 nm and a molecular mass range from ca. 2,000 Da to hundreds of MDa.
- Arbitrary solvents can be applied.
- A wide range of concentrations is accessible – formulations can be examined in their original state (depending on suitable instrumentation)
- Detection is most versatile due to multiple, synchronous optical systems.
- Complex mixtures are fractionated with high statistical reliability, as all sedimenting objects are detected.
- Combination of sedimentation and spectroscopic properties allows characterization by orthogonal information.
The basic physical property utilised in Analytical Ultracentrifugation is mass, or density, respectively. This is the fundamental difference giving the Analytical Ultracentrifugation a unique position among all other common methods of analysis.
Analytical Ultracentrifugation techniques
Click the corresponding picture for a brief introduction into the basic Analytical Ultracentrifugation techniques.
Here a summary on Analytical Ultracentrifugation technique, focusing on application.
Theoretical background is available on our scientific website under www.analytical-ultracentrifugation.com.