Sedimentation equilibrium is applied for the determination of molecular masses. This is a classical technique in Analytical Ultracentrifugation application; in biochemistry countless molecular masses of proteins have been measured with this experiment for decades. Nowadays, alternative and often less pricy methods are favoured, especially SEC, and in many cases, these methods are quicker as well.
Nevertheless, the following reasons still make Analytical Ultracentrifugation an attractive approach:
- In chromatographic methods, problems of column-sample interactions are encountered.
- In many cases, a significantly different eluent than the original solvent (formulation), is required, i.e. the result does not correspond to the original sample.
- No feasible standard may be available, of calibration is inefficient, calling for an absolute method.
- No column for the specific solvent is available.
- In other methods, particle geometries might produce misleading results.
- Interparticular interactions may be of interest – obscuring results with other methods, but identified and quantified by AUC.
- …
The accuracy of Analytical Ultracentrifugation equilibrium results (3% error) is unattained by other methods. The sample volumes needed for this experiment are extremely small.
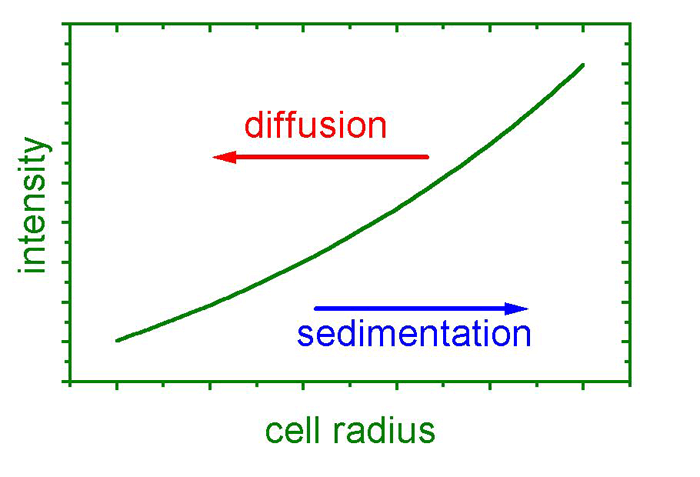
During a sedimentation equilibrium experiment, a dynamic equilibrium of sedimentation and diffusion is attained inside the sample cell. At first, the centrifugal field causes the material to sediment towards the cell bottom, increasing the local solute concentration. This causes back diffusion (diffusion is concentration dependent) to increase, opposing the centrifugal field. After sufficient time (this can take several days), a stationary condition is reached. The figure shows a typical exponential concentration profile in sedimentation-diffusion-equilibrium. From this profile, a weight-averaged molecular mass can be calculating, involving particle density and experimental conditions.
Molecular masses can also be obtained from sedimentation velocity experiments, which is much faster and has the advantage of yielding molecular mass distributions rather than mass averages. This evaluation, however, requires more or less complex global fits, including both sedimentation and diffusion processes. Other obstacles may be non-uniform frictional properties in mixtures and interparticle interactions. The complete hydrodynamic set of parameters is addressed, wheras for sedimentation equilibrium, the set is reduced to mass and density – excluding any properties connected to transport processes. This makes the equilibrium experiment more reliable when molecular mass is the actual target of interest.
For molcular mass distributions, the weight average is obtained, but numerical and z-averages are also accessible. This is a question of evaluation, and countless approaches have been documented in the literature. We use the M*-function developed by Creeth and Harding for evaluation. This is a rather sophisticated approach, requiring no model and providing high statistical certainty.
More detailed information on the theoretical background are compiled on our scientific website.

