The determination of ks, the concentration dependence of sedimentation, is essential für quantifying interparticular interactions in a system. This constant addresses interactions between sedimenting particles, mostly due to charge effects and shapes differing from spherical. Other effects, like the “hardness” or “softness” of a sphere can contribute to ks as well.
The evaluation of ks is often neccessary in advanced evaluations to eliminate concentration effects that might affect further results considerably.
But ks is not only an inevitable correction to account for strong interactions. This property is connected to others, such as intrinsic viscosity or the second osmotic virial coefficient, by scale relationships. These can be utilized to construct a full-scale hydrodynamic model of the particle in question.
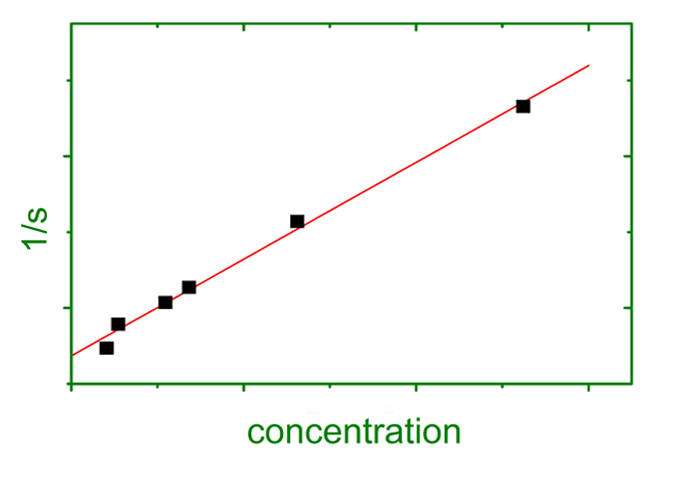
A series of six or seven concentrations is sufficient for a reliable determination of concentration dependence. First, sedimentation velocity is determined. A reciprocal plot of mean sedimentation constants versus concentration yields ks. Furthermore, the sedimentation constant at infinite dilution is obtained, a valuable parameter for hydrodynamic calculations.
Running a concentration series rather than a single sample raises expenses. Concsiderations in favour of the higher effort and cost could be the following:
- ks contains interesting information on particle properties, it is more than a (sometimes) necessary correction.
- The experiments required for ks determination can be otherwise evaluated, for example, for size distributions or density variation.
- Analytical Ultracentrifugation is an absolute method. This means that some effects must be corrected for, making such measurements neccessary – but on the other hand, no calibration is required.
Often enough, ks correction may be omitted. For instance, this is mostly the case when dealing with rigid spheres or with rather diluted samples.
More theoretical background can be found on our scientific website under www.analytical-ultracentrifugation.com.

